
For medical imaging technology, AI time has already arrived.
Seven years ago, there were almost no AI-enabled devices being approved. Since then, they’ve grown roughly in a straight line. Now about 40% of all radiology devices being approved are AI enabled, as the first chart below shows.
As a result, sales of AI diagnostic devices are likely to grow rapidly. The consensus forecast is for 33% compound growth over the next five years.
This means there’s a big opportunity for start-ups and established players. In some areas, AI has now become a necessary cost of doing business. In others it is possible to carve out a niche providing AI as a service particularly when serving the BioPharma industry.
The path won’t be completely smooth, however, according to the CGI report, because many doctors are reluctant to embrace AI-enabled devices. The full report has more on that.
AI-enabled devices as a % of all radiology devices approved by the FDA
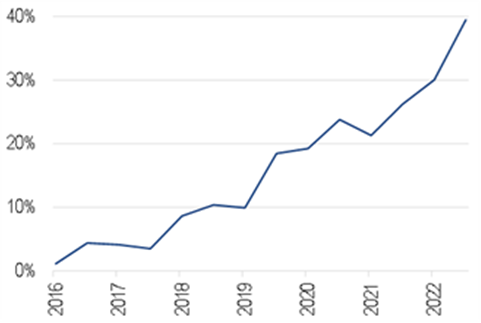
Source: FDA
Predictability of Reimbursement for Digital Devices

Source: BCG and UCLA Biobank
The FDA has tried to encourage innovation, and as a result AI-driven devices are now typically launched in the U.S.
By contrast the EU’s new safety-first approach has made it much harder to introduce products there. Many companies now say they will delay their European launches, if they do so at all.
In the U.S. the most important barrier to the use of new types of medical devices now revolves around “reimbursement” rather than “approval”. The FDA, or its equivalents overseas, may approve an AI-enabled device, but health providers will buy it only if they believe payers will reimburse its use. Unfortunately, the second chart above shows companies find it extremely hard to predict whether there will be reimbursement for new digital medical devices, both in the U.S. and in Europe.
Despite these issues, however, AI-enabled medical devices are growing rapidly – which means MedTech is likely to be the first area within health where AI will have a significant impact.
The number of AI-enabled medical devices approved by the FDA has grown steadily, as the first chart below shows, from close to zero before 2015 to 72 in 2H21 (the last six-month period we have full data for).
The growth is impressive, and there is no sign that the rate increase will slow down. However it’s important to put it in context: the second chart below shows that AI-enabled devices still represent only about 5% of all Class II medical devices being approved.
Figure 7. Number of AI devices approved by FDA (# per 6-month period)
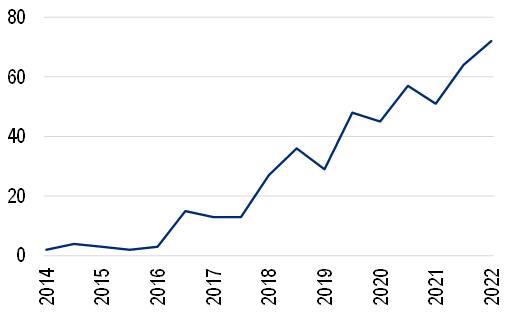
Source: FDA
Figure 8. AI-enabled device approvals as a % of all FDA Class II approvals
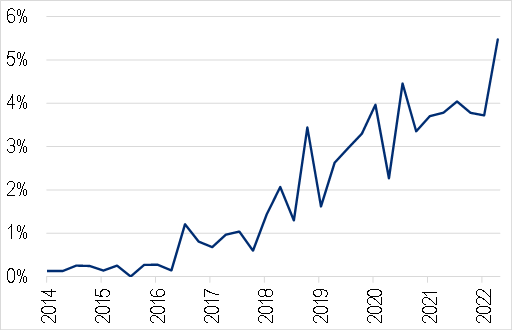
Source: FDA
About three quarters of all the AI products that have been approved are in radiology, in other words in medical imaging, with cardiovascular devices a distant second.
Almost 90% of those radiology devices are for diagnosis and that the main techniques in the field – CT, Ultrasound, MRI, X-Ray – are all well represented. Only 11% of AI-enabled radiology devices are dedicated to the therapeutic side of radiology – which typically involves using radiation to kill cancer cells, often in the earlier stages of cancer.
The full report looks in greater depth at how AI is being monetized in healthcare and covers several examples and case studies of AI in action across the sector.
To read it in full, if you are a Velocity subscriber, please see Smart Thinking on AI in Healthcare: Part 2 – Medical Devices: AI is increasingly disrupting radiology (6 April)
Citi Global Insights (CGI) is Citi’s premier non-independent thought leadership curation. It is not investment research; however, it may contain thematic content previously expressed in an Independent Research report. For the full CGI disclosure, click here.